 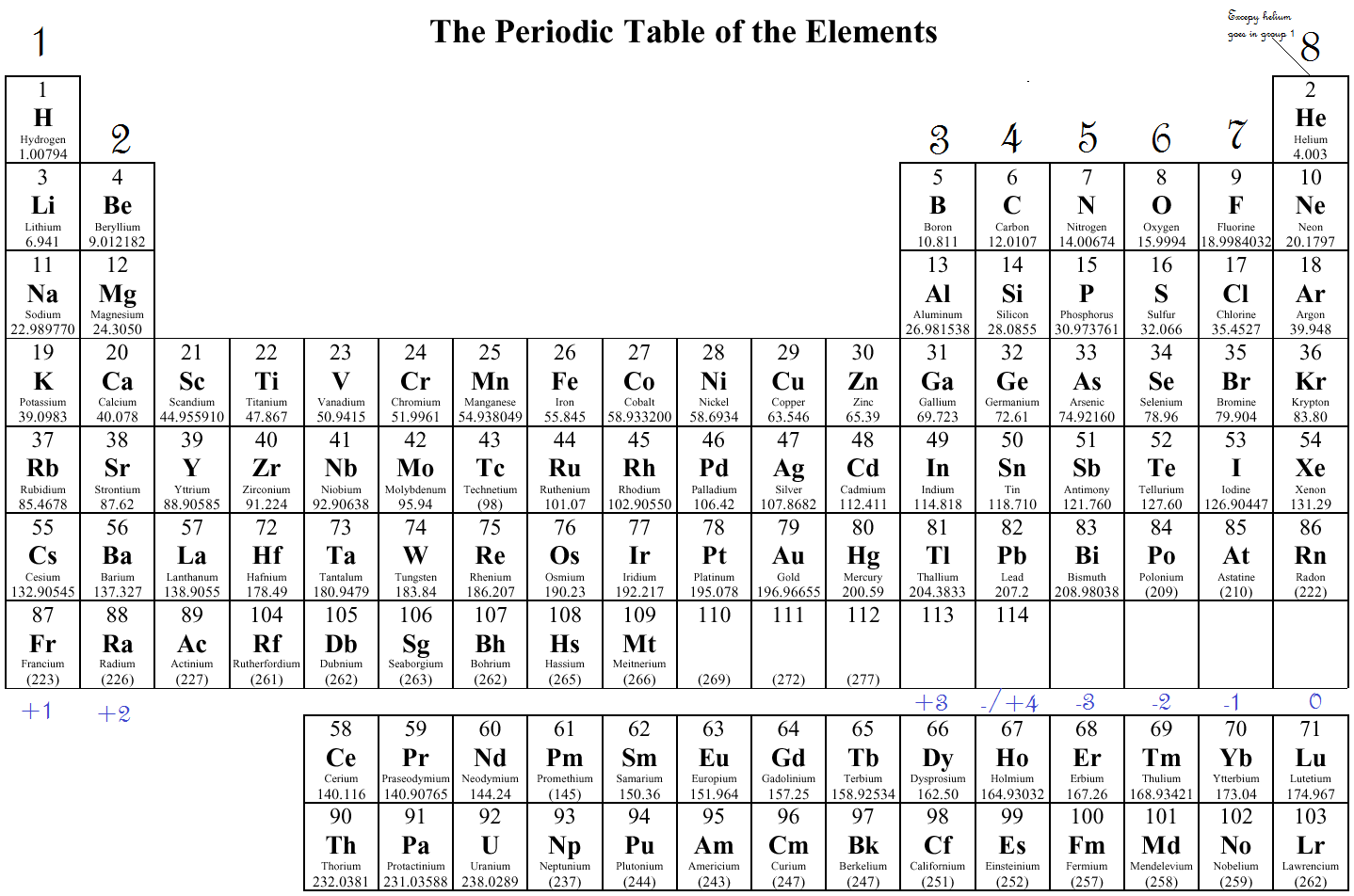
Or you can just remember that the transition metals are represented as groups 3 to 12. 
So just remember you could do with long way where you give the electron configurations of each transition metal and look at the number of total s and the electrons. There is also a 2019 edition of this table. Here is the pdf file of the color periodic table so you can save and print it. It includes element names, symbols, atomic numbers, atomic weights, element groups, and periods. OK, so just remember, if you're looking at the full out periodic table that I talked about in earlier chapters, you know that the transition metals are also represented as groups 3 to 12 and that new group number designation is also equal to the number of valence electrons. 2013 Edition This free periodic table wallpaper has a white background. Group Three A is group 13 all the way to group 18 for Group eight A. Group won a Group one group, Two Ways group, too. But they also have another set of numbers that describe them. You can use the periodic table to help you determine how many valence electrons an element (specifically, a neutral atom of the element) has. 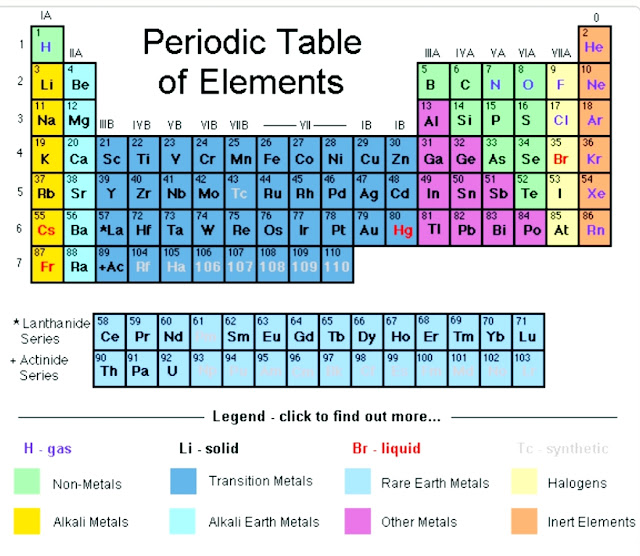
Remember our main group elements groups one a to eight a. This table shows the pattern in the periodic table that Mendeleev developed and how the missing elements at. This would be Group three Group 10, 11 and 12. Electrons orbit the atoms nucleus in energy levels. If you look at a full, out electric periodic table that we did in later in earlier chapters, you'll see that these groups also have another set of numbers for their group identity. Counting valence electrons for main group elements. Remember, besides classified transition metals is Group B elements. And then when we get to zinc, we see that it is two plus 10, which is 12. As a result, the most electronegative elements are found on the top right of the periodic table, while the. On the periodic table, electronegativity generally increases as you move from left to right across a period and decreases as you move down a group. If we look at them, this is two plus one, which is three, two plus two, which is four, and it goes on and on. Electronegativity is a measure of an atom's ability to attract shared electrons to itself. I've listed the outermost s orbital's and a deep orbital electrons for each of the transition metals. And here remember your transition Metal elements are known as your group B elements, so we know there's three b all the way to one Be all the way to eight b, but you don't look at that to determine the number of valence electrons. The two elements following argon in the periodic table are potassium, with a single 4s electron, and calcium, with two 4s electrons. Now with transition metals were going to say the number of Valence electrons equals their outermost s orbital electrons, plus their deep orbital electrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
